In the mid-1300s, fleas hitching rides on rats helped to set off the deadliest pandemic in human history. The rodents, infected with bubonic plague, had climbed aboard merchant ships and caravans heading from Asia to Europe — where, historians believe, the fleas abandoned the dying rats and moved in with humans. The infected bugs are cited as a major cause of the Black Death, which killed an estimated 75 million to 200 million people.
Nearly seven centuries later, another fatal illness that appears to have jumped from animals to people spread more quickly on the efficient wheels of modern travel. While the bubonic plague took weeks (at least) to reach neighboring regions from Asia on the sluggish transportation of the Middle Ages, the COVID-19 virus whisked across continents and oceans from China in days, riding with people on planes, trains, and automobiles. It ranks as the sixth most deadly human epidemic or pandemic in history, claiming 5.4 million lives and counting.
COVID-19 is the latest disease to demonstrate an alarming trend in infections: Zoonoses — diseases that spill from animals to humans — are occurring more frequently and spreading faster than ever. Zika, swine flu, West Nile virus, severe acute respiratory syndrome (SARS), and Middle East respiratory syndrome are just some of the major zoonotic epidemics and pandemics from the past several decades. The United Nations Environment Programme (UNEP) estimated in 2016 that up to 75% of emerging infectious diseases in humans are zoonotic.
“It’s going to happen more often,” says David Morens, MD, senior advisor to the director
at the National Institute of Allergy and Infectious Diseases (NIAID), based in Bethesda, Maryland.
That’s not because viruses are getting stronger; rather, infectious disease experts say, human behavior has increased conditions for people to catch diseases from animals and accelerate the spread of infections, largely by bringing people and animals into more frequent contact through development and travel.
“The world is getting closer together,” says Jay Varma, MD, director of the new Center for Pandemic Prevention and Response at Weill Cornell Medicine Medical College in New York City.
The center is among a slew of recent initiatives designed to address the problem. Last year, NIAID began funding an $82 million grant program to create a global network of Centers for Research in Emerging Infectious Diseases (CREID), with an emphasis on zoonoses. This past May, several international organizations — including the UNEP and the World Health Organization — created a One Health High-Level Expert Panel to “improve understanding of how diseases with the potential to trigger pandemics, emerge and spread,” also with a focus on zoonoses. This fall, the School of Veterinary Medicine at the University of Pennsylvania (Penn Vet) in Philadelphia opened the Institute for Infectious and Zoonotic Diseases to foster innovative strategies with health researchers, wildlife management agencies, and others.
Among the strategies for all: Improve surveillance of animals to curtail and maybe even prevent the spread of zoonotic diseases.
“Preventing spillover [to humans] is the real way to prevent epidemics,” says Jonathan Epstein, DVM, PhD, MPH, vice president for science and outreach at EcoHealth Alliance, a nonprofit that leads a collaboration in the CREID Network to improve the understanding of and response to zoonotic outbreaks in Southeast Asia. But because prevention involves so many complicated strategies, “that’s the hardest thing to do.”
Animal germs jump to people
Animals and humans trade bacteria, parasites, and fungi all the time, usually to no harmful effect. As explained by the Centers for Disease Control and Prevention, people commonly contract animal germs through contact with infected creatures (typically with their bodily fluids or through a bite), time spent in areas where those creatures live (such as among chicken coops, caves, and collections of water), or consumption of contaminated food (such as fruit soiled by animals).
Among the challenges to preventing zoonoses is that their routes to humans can be direct or circuitous. The viruses that cause versions of swine flu, for example, jump from pigs to humans — mostly at farms, researchers believe. Other zoonoses are delivered by so-called vector insects, which transfer pathogens from host animals to people. These illnesses include Zika (from monkeys via mosquitos) and Lyme disease (from deer and mice via ticks). Some zoonoses use animals as intermediaries.
The COVID-19 pandemic has given scientists an opportunity to focus the world’s attention on how to protect against more pandemics. The reasons for the growing risk include the expansion of human development (such as suburban sprawl) and activity (such as deforestation) into the territories of wild animals; climate change, which is forcing animals to migrate into areas populated by people; the globalization of trade, including of animals and animal meat for consumption; urbanization, which is squeezing people and animals into denser living conditions; and more frequent and speedy human travel around the world.
In summary, Morens says, “We’re stirring the pot.”
Some of the world’s most active pots are in Southeast Asia, where scientists frequently venture into animal habitats to track zoonotic outbreaks.
Surveillance as early warning
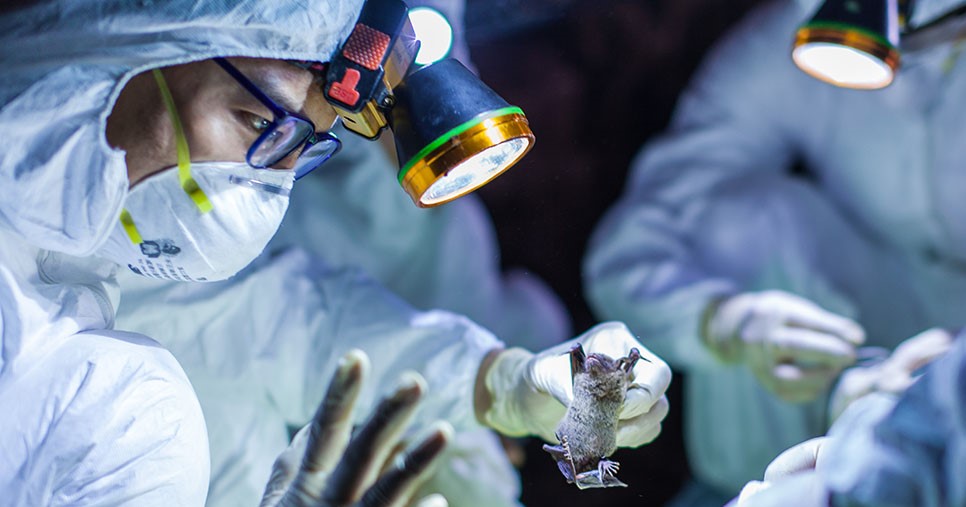
In Thailand, bats are ubiquitous around woodlands, waterways, farms, and homes; they’re even pitched as tourist attractions. Bats are also among the world’s most prolific culprits of zoonoses because they host lots of viruses that don’t sicken them but that they spread as they fly from place to place, biting and getting eaten by other creatures and dropping guano, which people harvest as fertilizer.
That’s why scientists spent two decades there (2001-20) collecting blood, urine, and nasal samples from fruit bats, pigs, and hospital patients to see if they carried the Nipah virus (NiV), a rare but deadly zoonotic disease that killed 100 people in neighboring Malaysia and Singapore in 1999 and keeps reemerging among humans in several countries. The pig and human samples in Thailand tested negative, but in 19 of the years the scientists found the virus in bats.
“The risk of a NiV outbreak in Thailand is increasingly possible,” the researchers warned in a report published last July.
Animal surveillance is a growing strategy to detect the spread of pathogens from one species to the other. The process is routine among livestock used in food production — to get an early jump on diseases that might wipe out animals as well as infect people — but scattershot among wildlife, due to the effort and cost of getting to habitats and collecting samples. The scientists in Thailand, for example, visited farms to swab mucus from pig noses and forests to draw blood from bat wings and lay tarp under trees to catch bat urine — all to track a potential outbreak.
Putting in that effort for an uncertain return is why preventive surveillance is not the norm. Governments and universities typically launch surveillance after a patient is stricken by an illness suspected of coming from an animal — because the virus is unknown among humans or has been found in animals before. The detective work includes determining what animals and animal spaces the patient had been in contact with and searching genetic databases kept by universities and governments to see if the pathogen in the patient matches any that have been found in animals.
“You want to get an idea of where the problem is most likely to be coming from, then do more close-up surveillance of animals and humans in that area to hone in on the hot spots” where transmission is most likely occurring, Morens says.
Once hot spots are found, mitigation actions include continuously testing people and animals to track the contagion; improving human sanitation practices; minimizing human contact with species that host the pathogen (such as by not consuming the host animal and not entering its habitats); and, as a last and controversial resort, killing off thousands of the host animals.
During the project in Thailand, government and academic institutions launched a campaign — based on a book, Living Safely with Bats, developed by the U.S. Agency for International Development — to teach people how to protect themselves. The strategies included not killing, cooking, or eating bats (which is common in parts of Asia) and not drinking water that might include bat droppings.
Sampling animals also increases knowledge about how a pathogen works. “Understanding these viruses gives us the ability to inform the development of drugs and vaccines, to know what other related viruses are out there, and to more rapidly trace outbreaks,” says Epstein at EcoHealth, which is based in New York City.
He adds, however, that animal surveillance — as it’s currently carried out — has significant limitations.
Future of surveillance
Early research indicated that Ebola was transmitted to humans by apes — maybe. And that SARS was transmitted to humans by civets — maybe. Later evidence pointed to bats as the “natural reservoir.” One limitation of animal surveillance and genetic sequencing of viruses is that they cannot always determine precisely how a pathogen spilled over to people.
Another drawback is the after-the-fact nature of all responses to zoonoses.
“That’s the traditional paradigm: Wait until there’s a human outbreak, then put intervention into place,” Epstein says. “Look what happened with COVID. By the time we recognized a handful of cases, it was too late.”
He and other veterinary leaders advocate for more surveillance to be regularly carried out in places with high concentrations of animals that harbor viruses that might infect humans and where people come in frequent contact with them. Scientists could see what known and potential zoonoses are spreading among animals and monitor humans more closely, as was done in Thailand.
“We need to be thinking about doing that in areas where spillover events are quite possible, like wet markets,” notes Daniel Beiting, PhD, associate director of Penn Vet’s new Institute for Infectious and Zoonotic Diseases.
That approach has limits, too: It’s impossible to predict what virus or bacteria from an animal might infect people. “I cannot take a sequence of a virus that came out of an animal and tell you that it is definitely going to be a human pathogen,” says W. Ian Lipkin, MD, director of the Center for Infection and Immunity at the Columbia University Mailman School of Public Health in New York City.
That’s one reason that surveillance is just one of several strategies against zoonoses. Epstein and others who follow the One Health approach — which emphasizes managing the shared environments of people, animals, and plants — advocate for larger changes in human behavior, such as curbing development into areas heavily populated by wildlife, reducing deforestation, confronting climate change, and reducing consumption of certain animals.
At Weill Cornell Medicine, Varma sees academic medicine playing a larger role in these efforts, including increasing coordination among veterinary, academic medicine, and public health institutions to share data; providing more interdisciplinary training in medical and veterinary education to increase understanding of contagion; and helping physicians know when to ask ill patients about their contacts with animals that might spread disease.
Says Varma: “Doing cross-education about the role of environmental change in the emergence and transmission of disease, understanding how diseases emerge in animals and how human and animal health is integrated — building that into the training of clinicians will, over time, help to build the cultural change that leads to better protection for everyone.”
