The relationship between academia and industry — though necessary for innovation — can pose a slew of ethical dilemmas. The AAMC’s Heather Pierce, JD, takes a deep dive into conflict of interest and Convey®, an innovative tool that streamlines disclosures.
On Feb. 12, 2019, dozens of biomedical research experts met at the AAMC for an invitation-only symposium to discuss the complicated issues surrounding the disclosure of financial interests to biomedical journals.
AAMCNews spoke to Heather Pierce, JD, senior director of science policy and regulatory counsel at the AAMC, about some of the issues that brought the group together: why conflict of interest is so complicated, what’s being done to improve transparency, and the tools available to make the disclosure process more seamless.
Q: Let’s talk a bit about conflicts of interest. Why would physicians or institutions even consider a relationship with industry?
A: The review and management of conflicts of interest is important because, in many cases, the academia–industry relationship is beneficial to science and to patients.
If the right answer was an absolute firewall between industry and academia, that would be very easy. You would just pick: Do you want to work for industry or do you want to work for academia? But generally, only industry makes drugs and devices, and we want the best thinking in academic medicine to be guiding what compounds are being studied, what drugs are being pursued, and what research is being conducted to ensure that the drugs that do get made are safe and effective. So there’s a necessary symbiotic relationship between the two. But the ways in which academia engages with industry are limited and managed to prevent a physician’s primary responsibilities — patient care, teaching, and research — from being compromised by a responsibility or loyalty to a company.
Q: Who polices that relationship tension?
A: It’s mostly the institutions, because it’s the institutional reputation that is at stake. There are robust systems and processes that have developed over many years to identify and manage conflicts of interest. Every federally funded institution has a policy that requires researchers to disclose all of their industry ties, and then the institution determines whether there is a conflict of interest. Institutions weigh all these considerations carefully. Is the individual doing research in an area that might impact a company that's paying him or her? Is the work someone is doing for a company about advancing science or is it about marketing? These are very different kinds of relationships and should be treated very differently.
Q: If institutions already collect this information, why aren’t industry ties always disclosed the same way in journals when publishing studies based on that research?
A: The disclosure process as it currently exists was called “fragmented and burdensome” by the Institute of Medicine (IOM) in its 2012 discussion paper, Harmonizing Reporting on Potential Conflicts of Interest. Disclosure processes are institution-specific, and they don’t guarantee that what a physician is telling his academic institution is the same information he is disclosing to the journals that are publishing his work.
Variations in disclosure time frames, threshold dollar amounts, instructions on disclosure of “relevant” financial interests, and human error in a repetitive process of filling out different disclosure forms with each manuscript submission — sometimes dozens a year — can all contribute to differences in what gets disclosed. There are also publicly available sources of information about industry payments, such as the CMS Open Payments database. With every new source of information comes greater transparency, but also a greater likelihood of mismatches between information in different places, and that is a threat to public trust in the truthfulness of any of the information.
Q: After the IOM issued its 2009 report, Conflict of Interest in Medical Research, Education, and Practice, it convened a group of 40 stakeholders, including representatives from respected medical journals and academic institutions, to discuss the development of a disclosure collection platform. At its conclusion, the AAMC was asked to build the platform, and the resulting product is Convey®. How does it work?
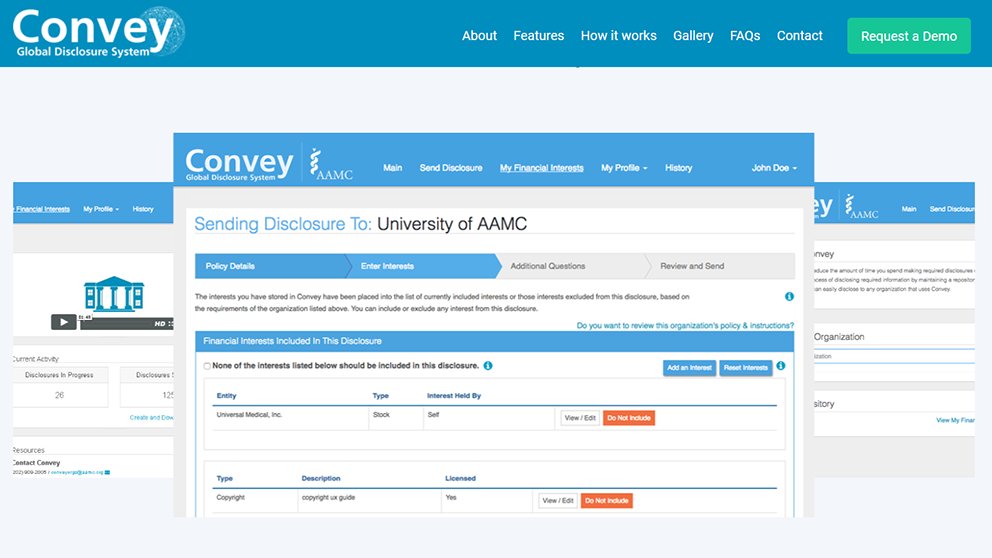
A: Convey® allows anyone to store all of their industry relationships in one place. After you log in to the free account, you input those interests to create disclosures for any organization that uses Convey — journals, academic institutions, continuing medical education (CME) providers, or nonprofit boards. Once you have disclosed one time, perhaps to one journal, any of those interests you disclosed are saved for you. Then later, maybe you’re writing a manuscript for another journal and the journal staff say, we want you to tell us which relationships are relevant to this manuscript. So you go into Convey and choose which relationships are relevant to that manuscript and then send a tailored disclosure to that journal. Each organization can decide what information it needs in a disclosure, and then Convey will guide the disclosers through that process. The platform itself is very flexible. Even if every organization has completely different requirements, Convey can accommodate that. And if in the future there is increased harmonization of requirements, Convey can use exactly the same standards for every journal or every CME provider.
Q: If everyone disclosed their industry ties through Convey, would it solve the problem?
A: Convey makes it easier to do the right thing, but it doesn’t solve the problem entirely. If an investigator wants to hide ties with industry, then Convey doesn’t fix that, but it can be a tool to compare public sources of information.
One important difference from the current system where a person fills out each form separately is that the Convey system makes it easier to disclose than not to disclose through an “opt out” process. Convey begins with the assumption that everything should be disclosed, and then the disclosers are asked to specifically exclude interests that aren’t relevant to the disclosure. Erring on the side of over-disclosure is a welcome culture and process change, especially in the journal context.
Q: If I’m a researcher, I might be leery about entering my financial relationships into a national database. Has this been a concern?
A: One of the primary reasons that the AAMC was asked to build this system was our long history of maintaining large amounts of private data because of our administration of the MCAT® exam, the ERAS® program, and AMCAS®. The important thing to remember is that the financial interest data stored in Convey aren’t publicly browsable; Convey is a private repository. The user has control over all of his or her own disclosure information, what it contains, and where it is transmitted.
Q: What’s next for Convey?
A: The momentum for use of Convey is growing. An editorial in the New York Times from the editor-in-chief of the New England Journal of Medicine (NEJM) announced that NEJM would be moving to collecting all disclosures through Convey, and other journals are starting or completing their pilot use of Convey now. Teaching disclosures are being collected for faculty at Harvard Medical School, and the Cleveland Clinic and UW Health at the University of Wisconsin have also committed to moving forward with disclosures through Convey.
We’ve developed a number of templates that are currently being tested and we are hopeful that we’ll see broader adoption through these mechanisms in the coming months.
For more information about Convey, visit www.aamc.org/services/convey or email convey@aamc.org.
