COVID-19 has been all-consuming. For nearly two years, the world has been focused on the race for vaccines, the pressures on providers, the best testing protocols, and simply staying safe.
COVID-19 also slowed some research efforts, but scientists still managed to seek solutions for many other pressing concerns — Alzheimer’s disease, maternal mortality, and prostate cancer among them — that have bedeviled patients for decades.
Below are eight medical advances that may not have grabbed your attention but could ultimately improve the lives of millions.
Rolling MRIs into hospital rooms

Assessing a stroke demands a rapid, life-or-death assessment: Is the culprit a clot, which requires a blood thinner, or bleeding in the brain, which requires surgery? Now, a portable MRI device can help make that assessment right at a patient’s bedside — and in much less time than required by a trip to a standard machine.
The Swoop MRI — which was created with input from Yale Medicine in New Haven, Connecticut — received Food and Drug Administration (FDA) approval in August 2020 and is already at work in several U.S. hospitals.
The new portable machine offers many advantages over its massive cousin, says Yale neurologist Kevin Sheth, MD.
“The very strong magnets in regular MRIs bring a lot of challenges,” he explains. “You need intensive power and cooling, precautions like a shielded room, and a lot of training. If you use a weaker magnet, all those problems go away.”
The weaker magnet is effective, according to an August 2021 study, which asked clinicians to identify various cerebral pathologies using Swoop images. “The goal is not to be as good as a high-magnet MRI, but to be good enough for clinical decisions,” says Sheth, who co-authored the study but has no financial interest in Hyperfine, the Connecticut-based company that produces the machine.
Swoop’s size — it’s smaller than some refrigerators — eliminates the need to move frail patients down hospital hallways. What’s more, its cost — around $100,000 compared to $1 million for the bigger machine — puts it within reach of hospitals and regions with fewer resources. “This could essentially democratize brain imaging,” argues Sheth.
Zapping prostate cancer
Prostate cancer strikes 1 out of 8 U.S. men, and it is expected to take more than 34,000 lives this year alone. When it metastasizes, the disease is almost always incurable, leaving physicians focused only on postponing death and improving patients’ lives.
A promising new approach has succeeded at both goals — and did so among men with an advanced form of the disease whose condition had deteriorated despite receiving standard treatments.
In fact, it more than doubled how long patients lived without their cancer worsening, according to a paper published in September. The study, which followed 831 men in 10 countries for a median of 20 months, compared patients who continued to receive standard care with ones who got the new treatment.
The treatment’s name is complex: lutetium-177-PSMA-617. But its approach is straightforward: Drive radiation directly into a cancer cell while sparing healthy tissue around it.
The method uses a compound called PSMA-617 to hone in on a protein found almost exclusively in prostate cancer cells, explains Oliver Sartor, MD, study co-lead investigator and medical director of Tulane Cancer Center in New Orleans. Then, a radioactive particle carried by the compound blasts the cancer cells, wherever they are.
“It’s like a little smart bomb,” says Sartor.
In September, the FDA granted the treatment priority review status, according to drug manufacturer Novartis, which funded the study. An answer is expected in the first half of 2022.
Sartor feels hopeful. “I’ve been working in prostate cancer for more than 30 years, and this is the largest advance I’ve ever been associated with.”
Solving sickle cell disease
For more than 5,000 years, sickle cell disease (SCD) has caused untold suffering in people of African descent. In patients with the genetic illness, red blood cells are not round but crescent-shaped — like a sickle — and can clog blood vessels, depriving the body of oxygen and causing tremendous pain. For a long time, the only cure has been a bone marrow transplant, but new gene-editing techniques now may offer a safe and effective alternative.
In research conducted at Boston Children’s Hospital, scientists used a virus to switch off the gene that triggers cells’ sickling, according to a January 2021 study. The patients subsequently produced healthy red blood cells — and nearly all were able to discontinue the blood transfusions SCD often requires.
“One participant used to have transfusions every month but has not needed any in three years,” says David Williams, MD, chief of the Division of Hematology/Oncology at Boston Children’s and head of the research team. “This has completely changed his life.”
The study followed six patients for a median of 18 months and found that the treatment completely halted the disease’s more severe symptoms.
“I’m so happy for my sickle-cell patients. This is a terrible disease,” notes Williams.
Next up for Williams is a trial with 25 patients. Meanwhile, SCD researchers elsewhere are studying other gene-editing techniques. “All these approaches look promising, and we need a lot more research to determine if one or another is better,” Williams says.
“This is a very exciting time. In the past, we haven’t had any particularly good treatments, and now we have several possibilities," he adds.
Saving mothers’ lives

When a woman’s uterus fails to contract after childbirth, tremendous blood loss can ensue, possibly leading to an emergency hysterectomy or even death. In fact, postpartum hemorrhage affects 3% to 10% of all childbirths in the United States and causes more than one-third of childbirth-related maternal deaths worldwide.
Treatment options include medications that don’t always work and inserting a balloon to put pressure on the uterus — much like exerting pressure on a cut — that comes with risks and must remain in place for a day.
But providers now have another option.
A new vacuum device aids natural post-birth contractions, putting pressure on leaking blood vessels. The FDA approved the device — the Jada vacuum uterine tamponade — in September 2020 following a 12-site research study.
“The vacuum approach is very logical since it’s like what the body is supposed to do,” says Dena Goffman, MD, the primary investigator at Columbia University Irving Medical Center in Manhattan. Also, the vacuum is used for less time than the balloon — roughly two or three hours. “For moms, that’s a big deal because it makes it easier to breastfeed, get out of bed, and bond with their child,” she adds.
The vacuum controlled bleeding in a median of three minutes and successfully treated 94% of participants, according to the study, which was funded by the device’s manufacturer, Alydia Health. In comparison, other research puts the balloon’s effectiveness at 87%.
“When a patient has a postpartum hemorrhage and you’re the doctor at the bedside, it’s scary because you know how quickly things can deteriorate,” says Goffman. “Using this device, when you see the bleeding slowing quickly and you can feel the uterus contracting, it’s just incredible.”
A reborn ACL
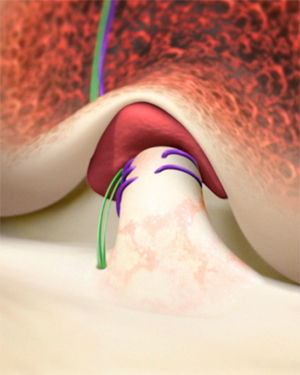
Tearing an anterior cruciate ligament (ACL) — the flexible band inside the knee that helps stabilize it — can upend a sports career and sideline weekend athletes. Between 100,000 and 200,000 ACL tears occur each year in the United States.
The most effective repair option has been removing the ruptured ACL, harvesting a graft from the shin or elsewhere, sewing that tissue into the knee, and hoping both surgical sites heal well.
In December 2020, the FDA approved a simpler, more natural method: the Bridge-Enhanced ACL Restoration (BEAR).
“We basically stimulate the ACL to heal itself,” says Martha Murray, MD, orthopedic surgeon-in-chief at Boston Children’s Hospital and BEAR’s creator.
The approach involves placing a protein-based sponge, prepared with some of the patient’s own blood, between the torn ACL ends. Murray explains that the blood promotes the connection of the two ACL pieces to the sponge and, ultimately, to each other.
So far, the approach has been tested on more than 100 patients. In a May 2020 study, patients and physicians reported that BEAR performed as well as the standard repair — and without the graft surgery that can cause ongoing pain or weakness at the donor site. Miach Orthopaedics, which has the worldwide exclusive license for the BEAR implant, has already begun making it available through orthopedic surgeons in the United States.
For Murray, the experience has highlighted the value of serving as a physician-researcher. “When you’re faced with a patient with a problem and the current solution is imperfect, it’s great to be able to say, ‘We’re working on a better solution.’ It’s incredibly gratifying.”
A breakthrough obesity medication
For the first time since 2014, a new obesity medication has hit the market, offering hope to the 78 million Americans who face the many risks of excess weight: cancer, heart disease, diabetes, and complications from COVID-19, among others.
And the new medication — semaglutide, also known as Wegovy — is significantly more powerful than its predecessors, according to research that helped it garner approval from the FDA in June.
“We’ve seen 1½ to 2 times the amount of weight loss compared to other medications,” says Robert Kushner, MD, a researcher at Northwestern University Feinberg School of Medicine who has led semaglutide studies. “That's a leapfrog advance.”
In fact, semaglutide recipients lost nearly 15% of their body weight on average — compared with 2.4% among controls, according to one study of nearly 2,000 patients.
Semaglutide — an injectable medication — is not entirely new. A synthetic version of a natural hormone that quells appetite, it’s already used to treat Type 2 diabetes. But the obesity trials, paid for by pharmaceutical company Novo Nordisk, used a much higher dose.
High doses haven’t been studied long enough to identify long-term side effects, notes Kushner, a paid consultant to Novo Nordisk. But the recent research reported mild-to-moderate gastrointestinal issues that lessened over time.
Now Kushner hopes semaglutide will help spark interest in obesity medications.
“Over 40% of U.S. adults have obesity, and the number who are getting a pharmacologic treatment is under 3%,” he says. “Part of the challenge is educating primary care providers that providing evidence-based obesity care includes consideration of medication."
The first blood test for Alzheimer’s disease
Randall Bateman, MD, a Washington University School of Medicine in St. Louis (WUSTL) neurologist, is thrilled to have contributed to the first blood test for Alzheimer's disease — a devastating condition that affects as many as 5.8 million Americans.
Back in 2017, though, as Bateman geared up to share the discovery that would enable the test, he worried about his peers’ reaction. After all, scientists were convinced that the blood marker he studied couldn’t predict the disease.
But the WUSTL method was much more sensitive and direct than prior approaches. The resultant test — called PrecivityAD — effectively detects the amyloid plaques that are a hallmark of Alzheimer’s disease and has proven as accurate as the previously used tools of a spinal tap or positron emission tomography (PET) scan, which are far more costly and complex.
The test, developed by a company called C2N Diagnostics that Bateman co-founded, has been available to physicians since October 2020, when it received approval through a federal lab certification program. It now awaits additional approval from the FDA.
“We’ve been hoping for a test to diagnose Alzheimer’s for more than 20 years,” says Bateman, WUSTL’s Charles F. and Joanne Knight distinguished professor of neurology. “Currently, up to half of people with Alzheimer’s are misdiagnosed.”
The road to success in science is paved with hard work and great uncertainty, he adds. “It’s a real gamble. You’re investing your life in this work, and you hope it will have a positive impact. And then it’s like, ‘Wow, it worked!’”
Ecstasy brings relief from severe PTSD
Anger, fear, recurring nightmares, and intense flashbacks are among the many symptoms that can batter patients with post-traumatic stress disorder (PTSD). The condition, which affects about 15 million U.S. adults in a given year, can be extremely difficult to treat.
A potentially groundbreaking PTSD treatment now lies in a seemingly unlikely source: MDMA, better known as the illegal drugs ecstasy and molly that fueled all-night dance raves and caused potentially fatal side effects.
In June, a study in Nature Medicine reported that patients with severe PTSD — combat veterans, first responders, and victims of sexual assault and mass shootings, among others — experienced significant relief from MDMA.
In fact, two months after treatment, 67% of subjects who received MDMA together with talk therapy no longer qualified for a diagnosis of PTSD. “I saw this amazing transformation in patients,” says Jennifer Mitchell, PhD, the study’s lead author and a University of California, San Francisco, School of Medicine neurology professor.
The treatment involved three eight-hour sessions a month apart during which patients ingested MDMA and processed painful memories and emotions in talk therapy.
MDMA releases a powerful supply of serotonin and stimulates hormones associated with emotional bonding, Mitchell explains. “The idea is that it helps patients be open in a way that enables them to connect well with therapists and work through their problems more quickly.”
Before the drug can receive FDA approval for PTSD, researchers need to complete one more clinical trial. Even if it succeeds, Mitchell is aware that MDMA still bears stigma from its party drug image.
“I hope people are going to be open-minded and look at the data,” which included no abuse potential or other serious side effects from MDMA as used in the study. “We are talking about use in a controlled, therapeutic situation,” she says. “Using drugs recreationally is entirely different. Otherwise, people would come back from [the art and community event] Burning Man cured of their psychological issues.”

