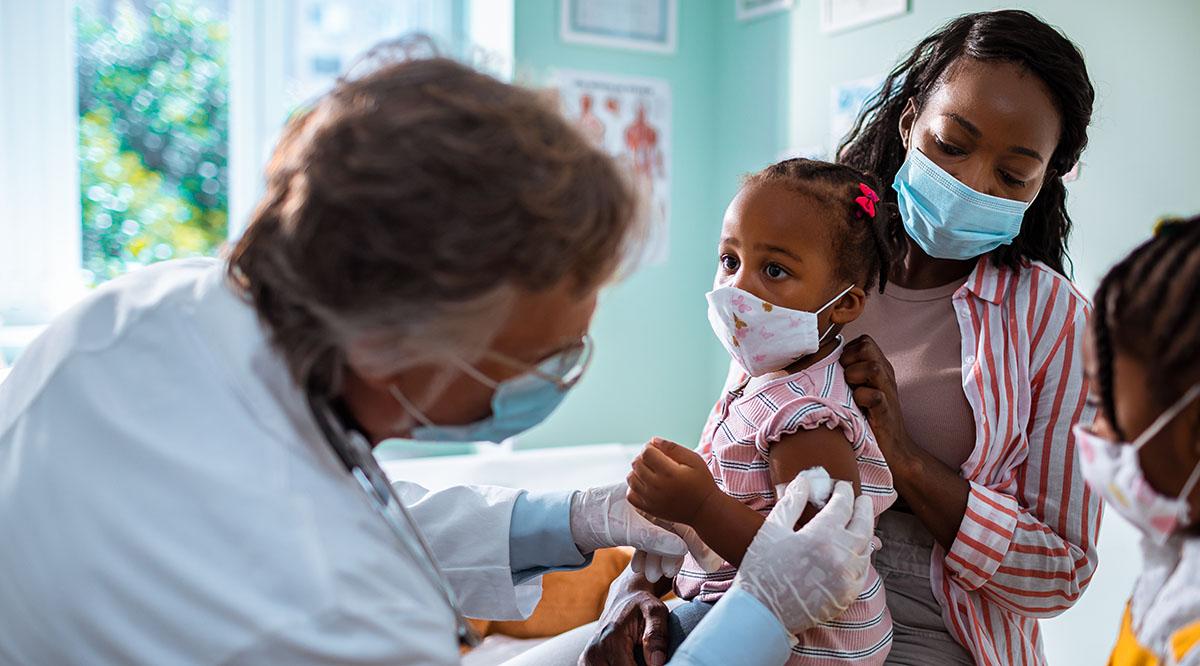
A few weeks ago, a small group of infants in Davis, California, received injections that millions of American parents wish their children could get: a vaccine to protect them from COVID-19.
But the immunization isn’t available to anyone else, because the infants aren’t humans — they’re monkeys. The 16 rhesus macaques at the California National Primate Research Center (CNPRC) comprise the only trial in the United States established to develop a COVID-19 vaccine for children. No one has started a human trial in the United States to produce a vaccine for kids, and no one can project when children might get vaccinated.
While Americans anxiously await the arrival of vaccines against the disease, many don’t realize that the immunizations being developed in the United States are intended for adults. A growing number of doctors and researchers are sounding alarms that the nation cannot beat COVID-19 until it inoculates children — and that if human trials for them don’t start soon, those inoculations might not be ready by the start of school next fall.
“A pediatric vaccine would not only help children — it will be the basis of eventually eliminating COVID-19 in our population,” says Sallie Permar, MD, PhD, an immunologist and professor at Duke University School of Medicine who co-leads the monkey trial with the CNPRC.
Permar and other researchers warn that holding off on developing a children’s vaccine risks the health of both children and adults, because immunizing children helps to curtail the spread of disease throughout their communities. The researchers point to how long it takes to develop, test, and approve vaccines for children, in part because their immunization responses can differ drastically from those of adults and among pediatric age groups.
“If pediatric trials don’t start soon, we’re going to lose our window” to vaccinate children by the start of the next school year, explains Evan Anderson, MD, a professor of pediatrics and medicine at Emory University School of Medicine in Atlanta.
The American Association of Pediatrics (AAP) joined the cause in a letter this month to the Food and Drug Administration (FDA). “It is counter to the ethical principle of distributive justice to allow children to take on great burdens during this pandemic but not have the opportunity to benefit from a vaccine … because they have not been included in vaccine trials,” the association said.
These voices cite the need not only to protect children from the disease but also to free them from home captivity for the good of their physical, intellectual, and emotional development — to let them return to classrooms, collective recreation, and companionship with each other.
“Kids don’t have anyone advocating for them and their need for a vaccine,” says Anderson, the lead author of a September article in the journal Clinical Infectious Diseases, “Warp Speed for COVID-19 Vaccines: Why are Children Stuck in Neutral?”
Little focus on kids
The child vaccine research advocates understand why COVID-19 efforts have prioritized adults: They have suffered the vast majority of known infections, severe illnesses, and deaths. The more than 697,600 COVID-19 cases that had been reported in children as of Oct. 8 accounted for 10.7% of all cases in the United States, according to an analysis of state data by the AAP and the Children’s Hospital Association. (The maximum age of those categorized as “children” varies from 14 to 19 among the states.) The 116 total child deaths by then were 0.06% of all COVID-19-related deaths nationwide, the analysis said.
In addition, the trials follow the FDA's guidance for COVID-19 vaccine development, which reflects the standard clinical trial process: Establish safety and efficacy in adults first, then move to children. Because of the widespread impact of COVID-19, though, many experts say we can’t wait until adult trials are complete.
To see why, James Campbell, MD, a professor of pediatrics at the University of Maryland School of Medicine (UMSOM), urges people to view the data differently: Imagine that adults were largely unaffected, he says, but over the past eight months the disease had infected more than half a million children, hospitalized thousands, and killed more than 100.
“Everyone would be saying this is the type of infection we’d want to have a vaccine for,” says Campbell, one of eight authors on “Why Are Children Stuck in Neutral?”
(For comparison, child deaths attributed to the flu tend to hover around 100 each season but fluctuate significantly. Fatalities dropped to 39 in 2011-12 and reached 189 in 2019-20, according to the Centers for Disease Control and Prevention [CDC].)
There has been some movement to insert youth into adult trials in the United States, which could accelerate the production of a vaccine for some children. Pfizer recently won FDA approval to include people as young as 12 in its RNA vaccine trial for adults. The company says it enrolled 16- and 17-year-olds under an earlier FDA approval, but won’t add anyone younger until it analyzes results from those older teenagers.
Other COVID-19 vaccine developers, including Johnson & Johnson, Moderna, and AstraZeneca, say they plan to run trials for children in the United States at some point. “Enrollment of children will start once sufficient data are gathered in adults,” AstraZeneca stated in reply to an inquiry. Several children’s vaccine trials are underway elsewhere, including the United Kingdom and China; the FDA could approve limited use of those in the United States.
Yet the CDC cautioned in a COVID-19 update on Oct. 14, “At first, COVID-19 vaccines may not be recommended for children.”
Child vaccines’ big impact
Children present an effective vaccination target for several reasons. For starters, there are a lot of them: The nation’s 73 million children (age 18 and under) account for one-fifth of its population, according to the U.S. Census Bureau. And another 3.7 million babies are born each year, creating what Anderson calls “a continuously growing group of unvaccinated people at risk for COVID-19 disease and transmission.”
Vaccinated children tend to produce stronger immune responses than adults do, and adult immune systems weaken considerably in old age, according to Koen Van Rompay, DVM, PhD, a research virologist at the CNPRC, which is part of the University of California, Davis. “If you can start at an early age, you set those children up for better long-term immunity,” says Van Rompay, who co-leads the monkey trial.
Consider the battle against measles, rubella, and polio: Those and several other diseases have been largely curtailed or essentially wiped out in the United States thanks to the federally recommended schedule of immunizations that begin in infancy. One reason for their success is that when doctors advise parents to immunize their children, parents comply: Completion rates for each of the recommended childhood vaccinations run from 80% to over 90%, according to the CDC.
Adults, on the other hand, show less enthusiasm about going in for their shots. Each year, less than half of adults get their federally recommended immunizations for flu, hepatitis, shingles, and more, according to the CDC. While a COVID-19 vaccine might draw more takers in the midst of the pandemic, widespread confusion about coronavirus research and political battles over the disease have eroded public confidence in scientific pronouncements about it. Recent polls show that about half of American adults say they will probably or definitely get a vaccine, while half say they will not.
Some researchers say that the routinely low immunization rates among adults means that immunizing only adults won’t achieve the widespread immunity that society needs to recover from the pandemic. What’s more, preliminary plans to first administer the vaccines to those at high risk of contracting or suffering from the disease (such as front-line health workers and people with certain health afflictions) would leave most of the nation’s population unprotected for the near future.
Vaccinations that first target groups of high-risk adults achieve good initial impacts within those groups, says Campbell at UMSOM, but the effect often flattens out because only a fraction of the target group gets protected, and just about no one else does. He cites vaccines against pneumococcus (which can lead to pneumonia and meningitis), Hepatitis A, and Hepatitis B as examples.
“They protect the people who get vaccinated, but they often fail on the bigger public health burden of reducing disease,” he says.
After those vaccinations were adopted into the routine schedule for children, infections dropped significantly in both children and adults, Campbell explains. He says unvaccinated children and adults get an indirect benefit from vaccines because the vaccines exponentially reduce how much virus is passed along in homes and communities: Parents, siblings, teachers, and friends don’t catch the virus from vaccinated kids — and don’t pass it along to others.
Immunizing millions of children will go a long way toward stopping the spread of COVID-19 among everyone, Van Rompay says: “If we can break the transmission cycle in children, we can help to protect the older population.”
A long process
The trouble is, developing a vaccine for kids can easily take most of a year or more, even at an expedited pace, researchers say.
After getting their pediatric trial plans approved by the FDA, developers typically proceed through steps by age group: They start with older children to determine dosage levels and frequencies that prove to be safe and effective, then they move down through younger age groups. At each step, researchers monitor the children to measure the vaccine effects, adjust the dosage amount and frequency as called for by the results, and then conduct more testing and monitoring. The vaccine candidates that successfully emerge from the trials go through an FDA review before approval for public use.
The first pediatric vaccinations might therefore go to older teens — who, among children and youth, appear to be the most likely to contract and suffer severe harm from the disease.
“You get into adolescence, and they really start to look like young adults as far as what their COVID-19 illness looks like,” says Matthew Kelly, MD, an assistant professor of pediatrics at Duke University Medical Center in North Carolina.
Kelly, who specializes in infectious diseases, says 6- to 13-year-olds “seem to have the lowest risk” of showing COVID-19 symptoms, and the severity of coronavirus infections appears to then increase among the youngest children. The CDC notes, “Babies under 1 year old might be more likely to have severe illness from COVID-19.” A vaccine for them might be produced last, depending on how the trials progress.
Although children’s vaccines must be tested in humans before going to market, vaccine developers can turn to nonhuman primates first or simultaneously. The trial with 3- to 4-month-old infant monkeys (equivalent to about 1-year-old humans) tests two vaccines, according to Van Rompay and Permar: an mRNA vaccine by Moderna and a protein vaccine from the Vaccine Research Center at the National Institutes of Health. The trial is run by the CNPRC, Duke, and the University of North Carolina.
The monkeys have received two vaccine injections, Permar says. The next steps, she explains, are to follow the “durability” of immune responses from the vaccines and, barring harmful immunization side effects, potentially administer small amounts of the novel coronavirus to see if the monkeys are protected from it.
If the vaccines prove to be safe and effective, Van Rompay says, “There will be much more confidence and trust [among vaccine developers and regulators] about getting these vaccines into human trials with infants and children.”